Diagnostic Tool for the treatment of diabetes in adults or children
Diagnostic Tool for the treatment of diabetes in adults or children
Identify a research or evidence-based article that focuses comprehensively on a specific intervention or new diagnostic tool for the treatment of diabetes in adults or children.
In a paper of 750-1,000 words, summarize the main idea of the research findings for a specific patient population. Research must include clinical findings that are current, thorough, and relevant to diabetes and the nursing practice.
ORDER NOW FOR AN ORIGINAL PAPER on Diagnostic Tool for the treatment of diabetes in adults or children
Prepare this assignment according to the APA An abstract is not required.
This assignment uses a grading rubric.
You are required to submit this assignment to TURNITIN
College of Nursing values honesty and integrity. All students should be aware of the Academic Integrity policy and follow it in all discussions and assignments.
By submitting this assignment, I pledge on my honor that all content contained is my own original work except as quoted and cited appropriately. I have not received any unauthorized assistance on this assignment. Please see the grading criteria and rubrics on this page.
Please see the grading criteria and rubrics on this page. Diagnostic Tool for the treatment of diabetes in adults or children
Diagnostic Tool for the treatment of diabetes in adults or children
The Pathophysiologic Basis for Diabetes Treatment: Evolving Perspectives , Presented by Lawrence Blonde, MD, FACP, FACE
The Growing Epidemic of Type 2 Diabetes
The last decade has seen a dramatic increase in the prevalence of obesity[1] which, in turn, has led to an associated increase in the prevalence of many obesity-related diseases, including diabetes. In fact, the increased prevalence of diabetes mirrors the increase in obesity during this period, helping diabetes to reach virtually epidemic proportions.
There now are 20.8 million Americans with diabetes, 20.6 million of whom are adults. This represents just under 10% of all adults in the United States. Among people 60 years of age and older — the fastest-growing age segment of our society — an alarming 21% have diabetes.[2] Type 2 diabetes is also being increasingly diagnosed in children and adolescents, at least in part as the result of an increase in obesity in that age group as well.
Diabetes is the number one cause of adult blindness, the number one cause of end-stage kidney disease, and the number one cause of nontraumatic amputation in this country. Diabetes also increases the prevalence of coronary heart disease and stroke by 2- to 4-fold. Largely related to these complications, direct medical and indirect expenditures attributable to diabetes in 2002 were estimated at $132 billion.[3]
Pathophysiology and Natural History of Type 2 Diabetes
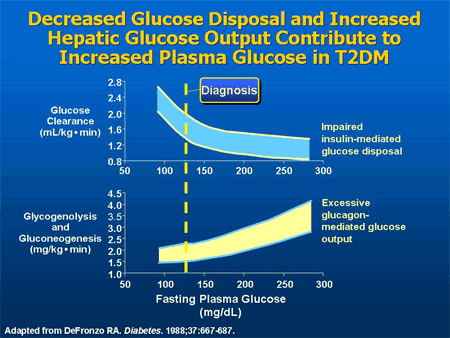
Most people who develop type 2 diabetes develop insulin resistance many years earlier. Initially, enhanced insulin production can compensate for the insulin resistance so that the person maintains normal fasting and postprandial glucoses. However, as insulin levels fall, there is first an increase in postprandial glucose levels followed by fasting hyperglycemia.[4]
Risk for microvascular complications of diabetes — the eye disease, the kidney disease — generally is associated with hyperglycemia sufficient to diagnosis type 2 diabetes. Macrovascular complication risks actually begin in the prediabetes stage, usually associated with insulin resistance. Moreover, many people have type 2 diabetes for 5 to 10 years before the diagnosis is made, so that a significant percentage of them already have complications at the time of diagnosis.[5,6]
The American Diabetes Association (ADA) identifies fasting plasma glucose (FPG) of less than 100 mg/dL as normal. A normal glucose value 2 hours after ingesting a 75-g glucose challenge is less than 140 mg/dL. Diabetes is defined as an FPG of 126 mg/dL or greater, or a 2-hour post-glucose challenge result of 200 mg/dL or greater. Fasting and/or post-glucose challenge values above normal but not diagnostic of diabetes are termed impaired fasting glucose (IFG) or impaired glucose tolerance (IGT), respectively.[3]
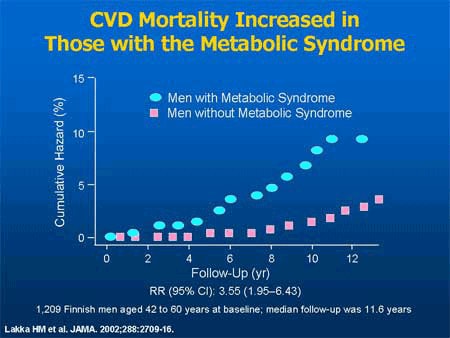
People with IFG, IGT, or both are said to have prediabetes. Prior to developing type 2 diabetes, most people have a prediabetes phase, and most people with prediabetes will progress to type 2 diabetes within about 10 years. However, even those who do not will have an increased risk of atherosclerosis.
Studies have indicated that type 2 diabetes can be delayed or even prevented. In the Diabetes Prevention Program study, approximately 3200 people with IGT were randomized into 3 groups. They either received a placebo, metformin, or a lifestyle intervention that had modest goals including losing at least 7% of body weight and exercising 30 minutes a day, 5 days a week, at the equivalent intensity of brisk walking. Compared with placebo, even this modest lifestyle modification was associated with a 58% reduction in the progression from prediabetes to diabetes.[7] So we know that we can at least delay, if not prevent, the progression from prediabetes to diabetes. Therefore, at-risk individuals should be screened during healthcare visits to identify those with pre-diabetes. This approach will also uncover a significant number of people with undiagnosed diabetes so they can receive needed treatment.
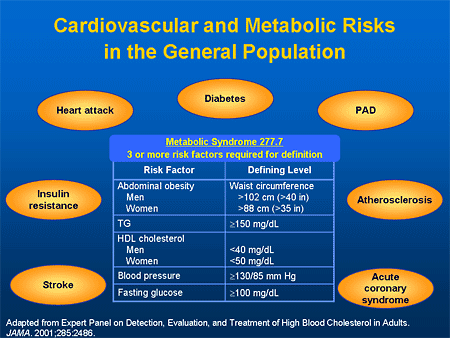
Insulin Resistance Syndrome and Metabolic Syndrome
Factors Contributing to Insulin Resistance
Insulin Resistance and Type 2 Diabetes
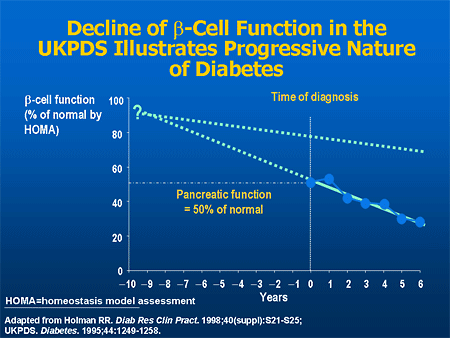
Studies of Beta-cell Mass
Autopsy studies comparing the volume of beta cells in nondiabetic individuals with that of people with diabetes found a 41% decrease in beta-cell mass among people with type 2 diabetes, regardless of whether individuals were lean or obese.[17] Another cadaver study that compared the islet-cell mass with the total mass of the pancreas revealed decreased islet-cell mass in individuals with type 2 diabetes compared with controls. Furthermore, glucose stimulation of isolated islet cells in vitro demonstrated that insulin release was decreased in cells from the patients with type 2 diabetes.[18] Progressive loss of beta-cell mass and beta-cell function, therefore, is an important therapeutic target in the management of people with type 2 diabetes.
Abnormal Insulin and Glucagon Response in Patients With Type 2 Diabetes
Administration of a bolus of intravenous (IV) glucose to a nondiabetic individual results in a biphasic insulin response: there is an immediate first-phase insulin response in the first few minutes, followed by a second-phase, more prolonged response. This first-phase insulin response is absent when a similar IV glucose bolus is administered to someone with type 2 diabetes. This deficiency contributes to the excessive and prolonged glucose rise after a meal in those with diabetes.[19]
In response to a carbohydrate-containing meal, individuals without diabetes not only increase insulin secretion but also simultaneously decrease pancreatic alpha-cell glucagon secretion. The decrease in glucagon is associated with a decrease in hepatic glucose production, and along with the insulin response, results in a very modest increase in postprandial glucose.[20]
In contrast, people with type 2 diabetes have a delayed and diminished insulin response to a carbohydrate meal, and their glucagon secretion is not decreased, and may even be paradoxically increased. These insulin and glucagon abnormalities produce an excessive postprandial glucose excursion. Historically, hyperglycemia in diabetes has been viewed as a failure of insulin-mediated glucose disposal into muscle and adipose tissue. However, these data suggest that hyperglycemia is also the result of excessive appearance of glucose due to unregulated hepatic glucose production. Infusing insulin can improve the insulin but not the glucagon profile. This is why, until recently, it has been difficult to control postprandial glucose levels in patients with diabetes. In fact, more than 30 years ago, Roger Unger presciently stated, “One wonders if the development of a pharmacologic means of suppressing glucagon to appropriate levels would increase the effectiveness of available treatments for diabetes.”[20]
The Role of Gastric Emptying in Postprandial Hyperglycemia
Another factor contributing to postprandial hyperglycemia is accelerated gastric emptying. The rate of gastric emptying correlates directly with postprandial glucose increments. Many factors, such as the glucose levels themselves, the form of the food ingested, etc, can affect the rate of gastric emptying. However, studies suggest that all other factors being equal, most people with type 1 and type 2 diabetes have accelerated gastric emptying compared to those without diabetes.[21,22]
The Role of Incretins in Glucose Homeostasis
In healthy individuals, an oral glucose load is associated with a greater insulin response than administration of an isoglycemic IV glucose infusion designed to mimic the plasma glucose excursion achieved by the oral glucose load. This enhanced insulin response to oral glucose has been called the incretin effect.[23] Incretin hormones were discovered when researchers tried to understand what caused this enhanced insulin response to oral glucose. These hormones are produced by the gastrointestinal tract in response to incoming nutrients, and have important actions that contribute to glucose homeostasis.
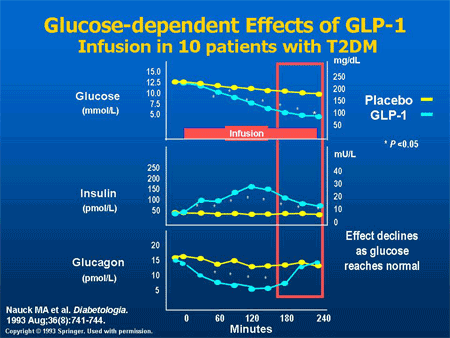
The 2 most important incretins are gastric inhibitory polypeptide (GIP) and glucagon-like peptide-1 (GLP-1). Type 2 diabetes patients have a resistance to GIP, making it a less attractive therapeutic target. GLP-1 is a 30-amino acid peptide secreted in response to the oral ingestion of nutrients by L cells, primarily in the ileum and colon. There are GLP-1 receptors in islet cells and in the central nervous system, among other places. GLP-1 is metabolized by the enzyme dipeptidyl peptidase-IV (DPP-IV), and the secretion and levels of GLP-1 are decreased in people with type 2 diabetes.
GLP-1 is responsible for a number of actions that help control glucose levels after a meal. It enhances glucose-dependent insulin secretion, inhibits glucagon secretion and therefore hepatic glucose production, and slows gastric emptying. GLP-1 also increases satiety resulting in less food intake. GLP-1 also appears to stimulate insulin gene transcription and insulin synthesis.
In animal studies, GLP-1 has also been shown to increase beta-cell mass by decreasing apoptosis or programmed beta-cell death, and increasing both beta-cell replication and neogenesis, where new beta-cells are formed from pancreatic ductal cells.[24]
Impact of Glucose Level Fluctuations
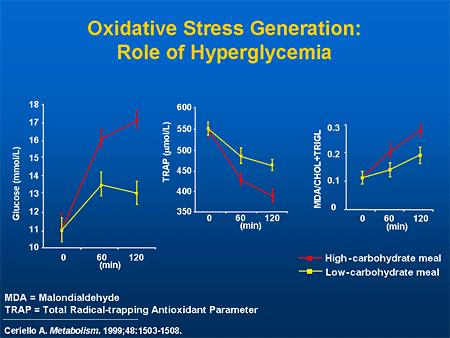
Diabetes is characterized not just by elevated fasting glucose, but also by elevated postprandial glucoses and enhanced variability of glycemia compared with the nondiabetic state.[26] An A1C of 7.5% can be the result of glucose levels that cluster about 175 mg/dL. However, an A1C of 7.5% can also be the result of glucose levels that vary from as high as 300 mg/dL to the low 50s. Therefore, while the A1C level is useful for assessing overall, average glycemic levels during the previous 2 to 3 months, it does not provide information about fluctuations or peaks in glycemia that may have harmful effects independently.
Reducing Cardiovascular Disease Risk Through More Aggressive Control
Numerous studies have demonstrated that lowering the A1C in both type 1 and type 2 patients reduces microvascular disease.[33-35] In the Diabetes Control and Complications Trial (DCCT), patients with type 1 diabetes were randomized to either 2 injections of insulin a day, or an intensive therapy consisting of 3 or more daily insulin injections or use of an insulin pump. During the course of this study, the people with more intensive insulin therapy had significantly lower A1C levels and about a 50% decrease in the risk of complications.[33]
The recent report from the DCCT-Epidemiology of Diabetes Interventions and Complications (EDIC) researchers demonstrated that many years after the study, type 1 patients assigned to intensive treatment in the DCCT had a 42% decrease in risk for any cardiovascular outcome and a 57% reduction in the risk for nonfatal myocardial infarction, stroke, or death from CVD, even though there was little difference in the A1C levels of the 2 groups during most of the follow-up period.[36] There are 2 important messages from this study: improving glycemia will reduce the risk for macrovascular disease, and while it is never too late to treat the hyperglycemia of diabetes, the earlier the treatment is begun, the greater the likely benefit because of an apparent metabolic memory of good and bad control.
Collepals.com Plagiarism Free Papers
Are you looking for custom essay writing service or even dissertation writing services? Just request for our write my paper service, and we'll match you with the best essay writer in your subject! With an exceptional team of professional academic experts in a wide range of subjects, we can guarantee you an unrivaled quality of custom-written papers.
Get ZERO PLAGIARISM, HUMAN WRITTEN ESSAYS
Why Hire Collepals.com writers to do your paper?
Quality- We are experienced and have access to ample research materials.
We write plagiarism Free Content
Confidential- We never share or sell your personal information to third parties.
Support-Chat with us today! We are always waiting to answer all your questions.